| Chapter 15: Alcohols, Diols and Thiols |
| Chapter 15: Alcohols, Diols and Thiols |
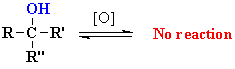
Oxidation of Alcohols
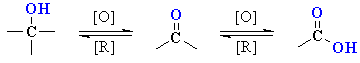
Reaction type: Oxidation-Reduction
Summary
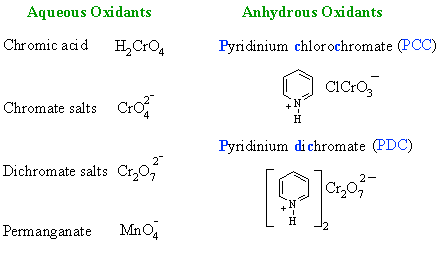
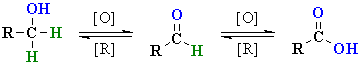
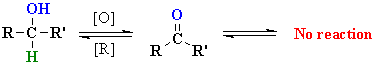
| Cr OXIDATION
OF ALCOHOLS |
|
| The mechanism is not trivial,
so attention here is focussed on the actual oxidation step. Prior to this,
the alcohol reacts to form a chromate ester (shown). A base (here
a water molecule) abstracts a proton from the chromate ester, the C=O
forms and a Cr species leaves. This demonstrates the importance of the carbinol H to this mechanism. |
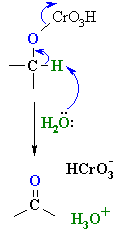 |
| Study Tip: If you see Cr reagents, you are probably looking at an oxidation reaction. Most likely the oxidation of an alcohol or an aldehyde |
| © Dr. Ian Hunt, Department of Chemistry |