Part 8: THERMODYNAMICS
This should have been a reasonably straight forward calculation, but care needs to be taken to get the right structures and the right molecular formula, then do the math correctly.
a. C6H12 + 9 O2 --> 6 CO2 + 6 H2O
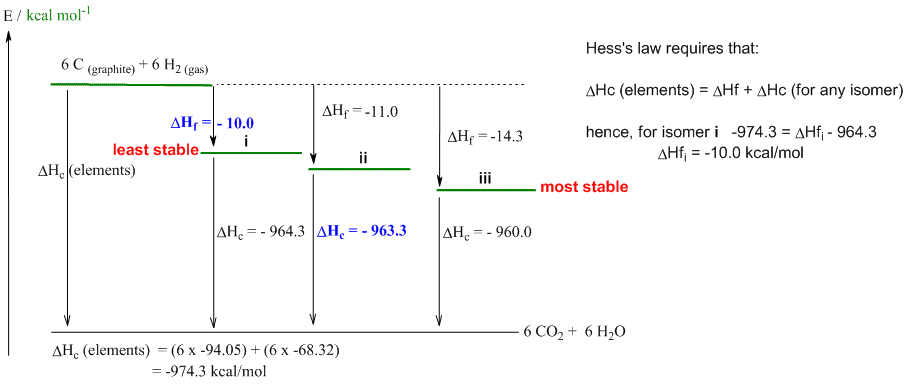
b. The calculation is shown on the figure below.
c.

d. See diagram above.
e. The difference between the 3 isomers is in the degree of branching. Hex-1-ene i is the least branched and the least stable (DHf = -10.0 kcal/mol) in contrast to isomer iii 3,3-dimethylbut-1-ene (DHf = -14.3 kcal/mol) which is more branched. A more branched isomer has more primary C-H bonds which are stronger than secondary C-H which in turn are stronger than tertiary C-H.
f.
![]()
Common errors:
In part a:
In part d :