A schematic of the solution is shown below. The information from the question is given in black. Deductions directly from this information are given in red. Points that provide potential key information are shown in green which leads to the structures which are linked via the blue arrows to show the path required to work them all out. There are, of course, other possible thought pathways.
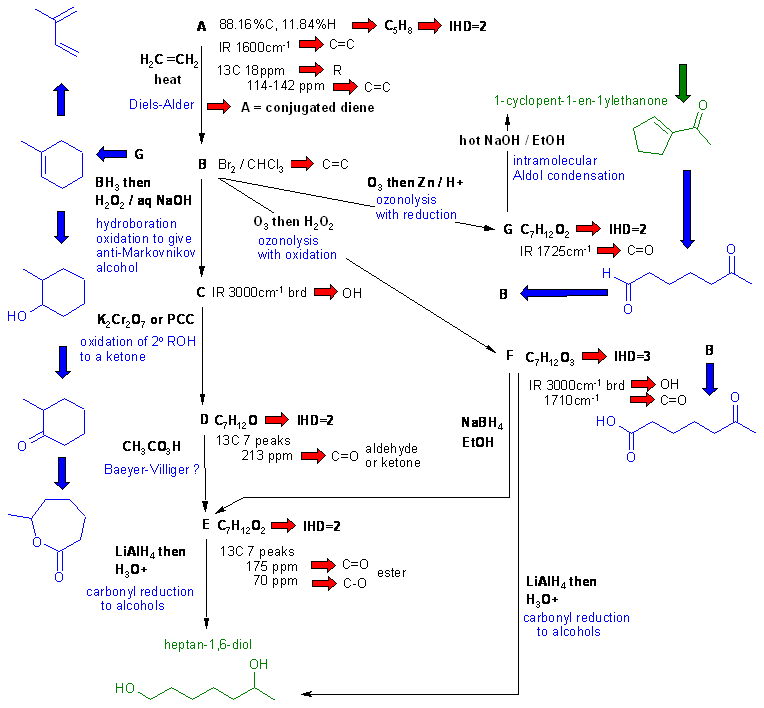
C, D and E contain chirality centers.
The key step into this problem seems to be the Aldol reaction by spotting the enone and reversing the intramolecular aldol to get G and then B from which you can also get F. B also provides access to A and C then D and E.
Alternatively, but a little more complex, once you get that A is a conjugated diene, C5, then there are only two possible structures, but one of those would give a Diels-Alder product as a racemic mixture... but B doesn't have chirality, hence giving the structure of A.
![[Chem 350 Home]](mol.gif)