
Note that these types of questions are often the most difficult on the examination. This is because they are holistic in that they require a good understanding of the reactions and the concepts and the ability to apply those concepts. They can't be memorised, they require understanding and ability to apply. Hence they help discriminate those who truly understand from those who have memorised and will soon forget.
A schematic of the solution is shown below. The information from the question is given in black. Deductions directly from this information are given in red. Points that provide potential key information are shown in green which leads to the structures which are linked via the blue arrows to show the path required to work them all out. There are, of course, other possible thought pathways.
Marking guide.... A - C 1.5 per structure, D - F = 2 marks per structure, 1 mark for the name of D.
Diagram 1 just gives all the information from the question in black with no interpretation. You can use this flow chart to try to solve the problem. Drawing a flow chart is a key step in developing the skills to solve these types of questions.

Diagram 2 gives the basic information from the question in black and deductions directly from this information are indicated by red arrows and with blue text plus it identifies the functional groups and points that provide potential key information by green arrows and key initial structures.
Diagram 3 gives the completed solution with the structures in blue. Key steps are the structure of the final product (based on the name and / or spectroscopic data) and the conjugated diene nature of C.
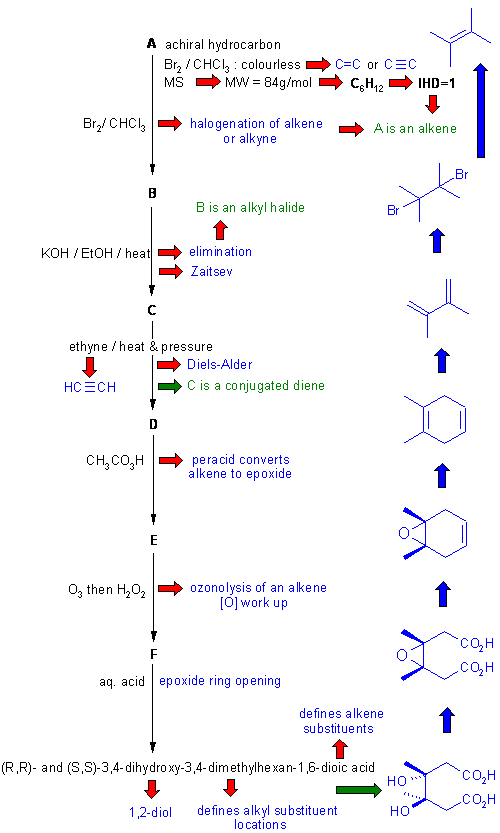
IUPAC name of D = 1,2-dimethyl-1,4-cyclohexadiene
The crux to this problem is recognising the reagents and the implications for the functional groups in the starting materials and products of each step. Working backwards from the named structure (also spectroscopic data) to get F and hence E then D. Note that the named structure defines the location of the alkyl groups through the whole series of compounds.
Recognising the F from the spectroscopic data is also important. Once D is determined, C, E, and F follow directly. C then gives B which in turn gives A. E allows access to H and also B, G is deduced from E and H from D. Note the how the symmetry and substitutent effects dictate the nitration reactions of D and E.
Common errors: (1) not checking the MW of the structure proposed for A. (2) poor interpretation of the reactions especially for A to B and C to D. (3) not naming or poor naming of D as asked. (4) guessing a structure of A then not checking other data.