Part
7: LABORATORY
Part A:
Experimental yields.... First you need to balance the reaction equation and then work out the moles of each reagent used to determine the limiting reagent:
| |
C6H5CO2CH3 + 2 CH3MgBr --> C6H5COH(CH3)2
|
| MW (g/mol) |
136.2 |
119.25 |
136.2 |
| amount (g) |
2.723 |
4.17 |
2.00 |
| mmoles |
20.0 |
35.0 |
14.7 |
| mmoles/ coefficient |
20.0 /1 = 20.0 |
35.0 / 2 = 17.5 |
14.7 / 1 = 14.7 |
Therefore, in the last row of the table where the stoichiometric coefficient from the balanced equation is divided into the number of moles, we can see that the limiting reagent in the Grignard reagent, methyl magnesium bromide. This means that the maximum amount of product that can be formed is 17.5 mmol (based on the reaction stoichiometry). Hence the % yield, based on obtained / max. possible = 14.7 / 17.5 = 84%.
Common general errors: (1) not balancing the reaction equation, (2) not determining the limiting reagent correctly and (3) not knowing how the calculate an experimental yield. This section was done poorly considering the number of times yields are calculated in the laboratory during the semester. Note that it doesn't matter whether you work with grams or moles of product the answer should be the same.
Part B:
- The molecular formula of C11H12O2 implies and IHD = 6.
- The solubility in 5% NaHCO3 suggests that we have a strongly acidic functional group.
From the chemical tests we get:
- 2,4-DNP test is negative and therefore indicates the absence of an aldehyde or ketone.
- The Tollen's test is negative confirms it's not an aldehyde.
- The ferric chloride test is negative indicating that it's not a phenol.
- The iodoform test is negative indicating that it's not a methyl ketone.
- The bromine test is positive indicating the presence of an alkene or alkyne.
The H NMR data tells us:
- 12.5 ppm is due to a carboxylic acid H (-CO2H)
- 7.5 to 8.0 ppm suggest 4 aromatic H and therefore probably a disubstituted benzene system.
- 6.4 ppm represents one H probably a vinyl H (i.e. sp2 C-H)
- 1.9 ppm shows a 6H singlet for two methyl substitutents.
The 13C NMR data tells us:
- 168 ppm is due to the carbonyl C in the carboxylic acid.
- 124-137 ppm suggest 8 sp2 C.
- 20 and 26 ppm represents 2 sp3 C in alkyl substitutents.
Summary....
Molecular formula of C11H12O2 implies and IHD = 6.
H NMR and functional group tests suggest -CO2H
NMR and functional groups test suggest trisubstituted alkene, C=C-H.
H NMR suggests a disubstituted benzene, C6H4 and 2 x CH3
Summing these pieces matches to the molecular formula and we have the IHD too (C=O, C=C plus 4 for the benzene system).
Altogether...
The fragments we have are : -CO2H, C=C-H, 2 x CH3 and C6H4 (i.e. disubs. benzene)
Assembling the pieces....
The aromatic H coupling pattern 7.5 to 8 ppm indicates a meta substitution (singlet, triplet and two doublets) rather than ortho or para.
The lack of coupling between the methyl groups and the vinyl H suggest that the methyl groups are 1,1- or geminal on the alkene i.e. C=C(CH3)2 |
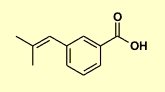
3-(2-methyl-1-propenyl)benzoic acid
|
The final step should always be to check what you
have drawn. The easiest thing to check is usually the coupling patterns you
would expect to see, and the chemical shifts of each unit. You should
be asking yourself : "Does my answer give me what the H-nmr shows ?"
![[Organic Home]](mol.gif) Return to Homepage
Return to Homepage

![[Organic Home]](mol.gif) Return to Homepage
Return to Homepage