 |
|||||
| amount (g) | 1.66 |
1.5 x 0.79 = 1.185 |
1.111 |
||
| mmoles | 10.0 |
25.7 |
5.0 |
||
| mmoles/ coefficient | 10.0 |
25.7 / 2 = 12.85 |
5.0 |
||
Part 7: LABORATORY
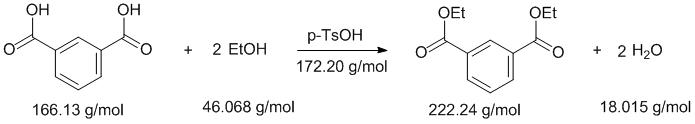
Experimental yields.... First you need to balance the reaction equation and then work out the moles of each reagent used to determine the limiting reagent:
Note that TsOH (tosic acid) is just a catalyst and chloroform is the reaction solvent and therefore can be ignored in terms of the yield calculation.
 |
|||||
| amount (g) | 1.66 |
1.5 x 0.79 = 1.185 |
1.111 |
||
| mmoles | 10.0 |
25.7 |
5.0 |
||
| mmoles/ coefficient | 10.0 |
25.7 / 2 = 12.85 |
5.0 |
||
Therefore, in the last row of the table where the stoichiometric coefficient from the balanced equation is divided into the number of moles, we can see that the limiting reagent in the phthalic acid (i.e. starting material). This means that the maximum amount of product that can be formed is 10 mmol (based on the reaction stoichiometry). Hence the % yield, based on obtained / max. possible = 5.0 / 10.0 = 50 %.
Common general errors: (1) not balancing the reaction equation, (2) not determining the moles of each material correctly (3) not determining the limiting reagent correctly (4) not knowing how the calculate an experimental yield and (5) reporting too many significant figures in the % yield. Note that it doesn't matter whether you work with grams or moles of product the answer is the same when done correctly.