| Chapter 9 : Alkynes |
| Chapter 9 : Alkynes |
Preparation of Alkynes via Elimination reactions
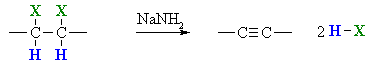
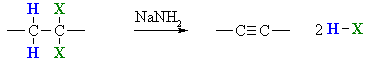
Summary
| What is the alkyne product from the reactions of the following with NaNH2: | |||
| (a) 2,2-dibromopropane | ANSWER | (c) 1,2-dibromohexane | ANSWER |
| (b) 1,1-dibromooctane | ANSWER | (d) 2,3-dibromohexane | ANSWER |
| © Dr. Ian Hunt, Department of Chemistry |