| Chapter 11 : Arenes and Aromaticity |
| Chapter 11 : Arenes and Aromaticity |
As a functional group, benzene, substituted benzenes and polybenzenes are called "arenes".
Nomenclature:
Functional group suffix = -benzene (review)
Functional group prefix = phenyl-
Substituted benzenes are usually named as such. The relative positions can be
denoted as 1,2- = ortho-, 1,3- = meta- and 1,4- = para-
substitution.
When polysubstituted, the numbers alone are used, e.g. 1,2,3-trimethylbenzene.
Many simple monosubstituted benzenes have common names that are still in use
- you will need to be familiar with these.
 |
 |
In benzene
all the CC bonds are known to be of equal length (above) so there are
no C=C and C-C. This is best represented by the resonance hybrid
in the Robinson form.
However, since the key to organic chemistry is being able to understand and draw mechansims
based on drawing curly arrows to account for the positions of the electrons, the
Kekule structures are strongly recommended as they give a more precise description of the electron positions
that can avoid confusion. Therefore, it is a good idea to use a Kekule representation.
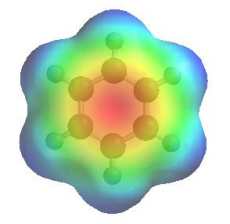 |
The image shows the electrostatic potential for benzene. The more red an area is, the higher the electron density and the more blue an area is, the lower the electron density. Note the nucleophilic character of the aromatic pi system. The reactivity issues can be separated into two types of reactions:
|
For reactions directly on the aromatic ring:
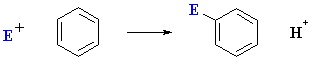
| © Dr. Ian Hunt, Department of Chemistry |