| Chapter 15: Alcohols, Diols and Thiols |
| Chapter 15: Alcohols, Diols and Thiols |
Synthesis of Esters
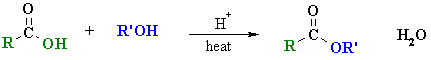
Summary
| Study Tip: The carboxylic acid and alcohol combination used to prepare an ester are reflected by the name of the ester, e.g. ethyl acetate (or ethyl ethanoate), CH3CO2CH2CH3 can be made from CH3CO2H, acetic acid (or ethanoic acid) and HOCH2CH3 (ethanol). |
This general "disconnection" is shown below:
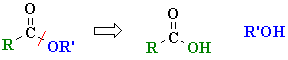
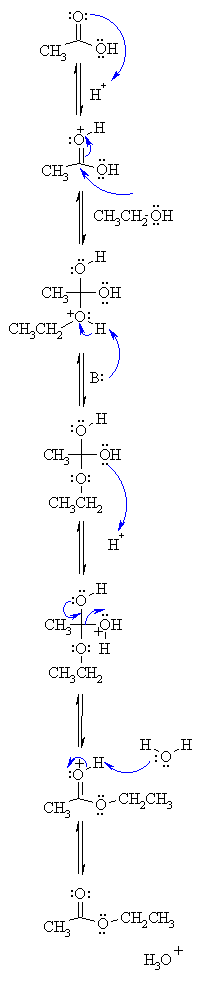
| MECHANISM
FOR REACTION FOR ACID CATALYSED ESTERIFICATION |
|
| Step 1: An acid/base reaction. Protonation of the carbonyl makes it more electrophilic. |
|
| Step 2:
The alcohol O functions as the nucleophile attacking the electrophilic C in the C=O, with the electrons moving towards the oxonium ion. |
|
| Step 3:
An acid/base reaction. Deprotonate the alcoholic oxygen. |
|
| Step 4:
An acid/base reaction. Need to make the -OH leave, so convert it into a good leaving group by protonation. |
|
| Step 5:
Use the electrons of an adjacent oxygen to help "push out" the leaving group, a water molecule. |
|
| Step 6:
An acid/base reaction. Deprotonation of the oxonium ion reveals the carbonyl in the ester product. |
|
| © Dr. Ian Hunt, Department of Chemistry |