| Chapter 16: Ethers, Epoxides and Sulfides |
| Chapter 16: Ethers, Epoxides and Sulfides |
SN2 type Reactions of Epoxides
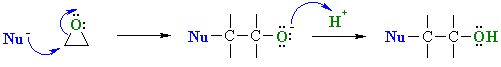
| Scenario 1: The Nu attacks the more substituted C of the epoxide at 180o to the C-O bond that breaks. |
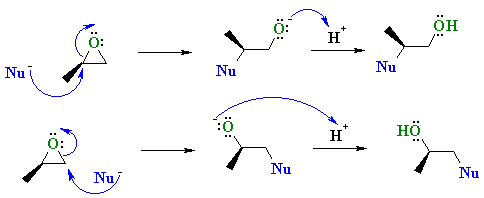 |
| Scenario 2: The Nu attacks the least hindered end of the epoxide at 180o to the C-O bond that breaks. |
| © Dr. Ian Hunt, Department of Chemistry |