| Chapter 17: Aldehydes and Ketones. Nucleophilic Additionto C=O |
| Chapter 17: Aldehydes and Ketones. Nucleophilic Additionto C=O |
Ozonolysis of Alkenes
(review of Chapter 6)
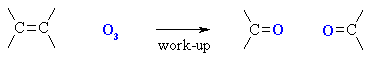
Summary
|
|
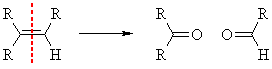
|
QUESTIONS What would be the products of the ozonolysis reactions of:
| ANSWER | (c) 2-butene ? | ANSWER | |||
| ANSWER | (d) 2-methylpropene ? | ANSWER |
MECHANISM FOR
REACTION OF ALKYNES WITH O3 |
|
| Step 1: The π electrons act as the nucleophile,attacking the ozone at the electrophilic terminal O. A second C-O is formedby the nucleophilic O attacking the other end of the C=C |
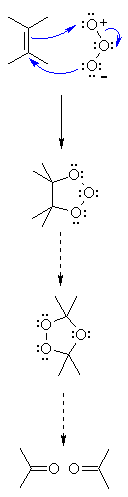 |
| Step 2: The cyclic species called the ozonide rearranges to the malozonide. |
|
| Step 3: On work-up (usually Zn / acetic acid) the malozonide decomposes togive two carbonyl groups. |
|
| © Dr. Ian Hunt, Department of Chemistry |