| Chapter 17: Aldehydes and Ketones. Nucleophilic Addition to C=O |
| Chapter 17: Aldehydes and Ketones. Nucleophilic Addition to C=O |
Hydride Reductions of
Aldehydes and Ketones
(review of Chapter 15)
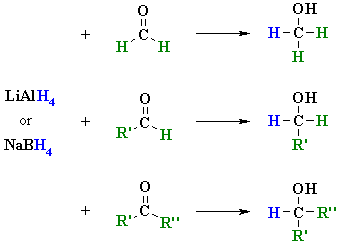
Reactions usually in Et2O or THF followed by H3O+
work-ups
Reaction type: Nucleophilic Addition
Summary
NUCLEOPHILIC
ADDITION OF LiAlH4 TO AN ALDEHYDE |
|
|
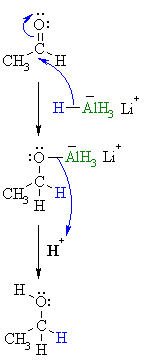 |
| Step 2: This is the work-up step, a simple acid/base reaction. Protonation of the alkoxide oxygen creates the primary alcohol product from the intermediate complex. |
|
| © Dr. Ian Hunt, Department of Chemistry |