| Chapter 17: Aldehydes and Ketones. Nucleophilic Addition to C=O |
| Chapter 17: Aldehydes and Ketones. Nucleophilic Addition to C=O |
Reactions of Primary Amines
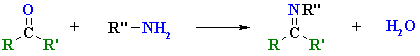 |
| imine |
Summary
Related Reactions
| NUCLEOPHILIC
ADDITION OF A PRIMARY AMINE GIVING AN IMINE |
|
| Step 1: An acid/base reaction. Protonation of the carbonyl activates it and makes it more susceptible to attack by a neutral nucleophilic like the N of a primary amine. |
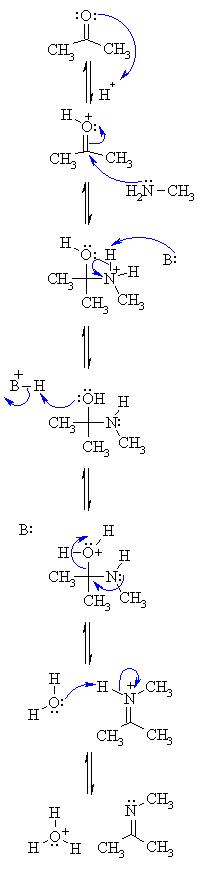
|
| Step 2: Attack of the N nucleophile at the electrophilic C of the C=O group with the electrons from the π bond going to the +ve O. |
|
| Step 3: An acid/base reaction. Removal of the proton neutralises the +ve charge on the N and forms the carbinolamine intermediate. |
|
| Step 4: To form the imine we need to dehydrate. However, before -OH leaves it needs to be protonated, so a simple acid/base reaction. |
|
| Step 5: Use the electrons of the N to help push out the leaving group, a neutral water molecule, forming a C=N in the form of an iminium ion. |
|
| Step 6: An acid/base reaction. Deprotonation of the iminium N reveals the imine product and regenerates the acid catalyst. |
|
| © Dr. Ian Hunt, Department of Chemistry |