|
|
|
|
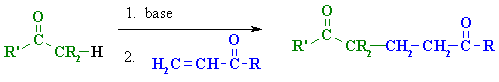
Summary
Related reactions
| Step 1: First, an acid-base reaction. Hydroxide functions as a base and removes the acidic α-hydrogen giving the reactive enolate. |
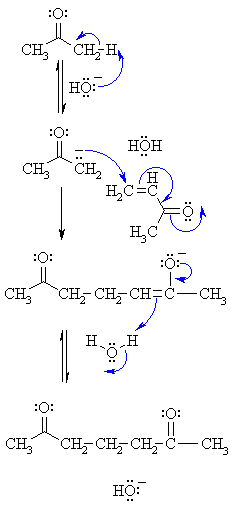 |
| Step 2: The nucleophilic enolate attacks the conjugated ketone at the electrophilic alkene C in a nucleophilic addition type process with the electrons being pushed through to the electronegative O, giving an intermediate enolate. |
|
| Step 3: An acid-base reaction. The enolate deprotonates a water molecule recreating hydroxide and the more favourable carbonyl group. |
|
| © Dr. Ian Hunt, Department of Chemistry |