 |
|
 |
|
| Nomenclature |
Formula |
3D structure |
| Functional class name = alkyl
alkyl ether e.g. ethyl methyl ether Substituent prefix = alkoxy- e.g. methoxyethane |
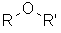 |
|
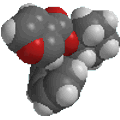
ethyl methyl ether
|
CH3CH2OCH3 |
|
|
CH3CH2OCH2CH3 |
|
1-methoxypropane *
|
CH3CH2CH2OCH3 |
|
2-methoxypropane *
|
CH3CH(OCH3)CH3 |
|
4-methoxybut-1-ene |
CH3OCH2CH2CH=CH2 |
|
* In reality, these examples are simple enough that they would typically be named as alkyl alkyl ethers |
||
|
CH3OCH2CH2CH3 |
|
|
CH3OC(CH3)3 |
|
| * In reality, these examples are simple enough that they would typically be named as alkyl alkyl ethers | ||
| ©Dr. Ian Hunt, Department of Chemistry |