Part 5: MECHANISMS
Note that no other reagents are needed in order to complete any of these sequences, you should only be using what is there.
General common errors:
(1) incorrect formal charges (2) backwards arrows (3) not showing the arrows for all the bonding changes (4) misuse of resonance / equilibrium arrows (5) vague arrows e.g. not starting where the electrons are i.e. on a bond or lone pair.
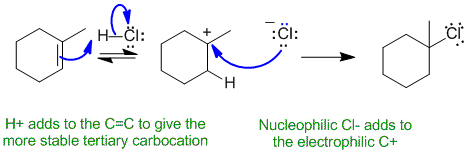
A1

Common errors : showing a radical reaction, a concerted reaction or a reaction via a less favourable secondary carbocation
A2
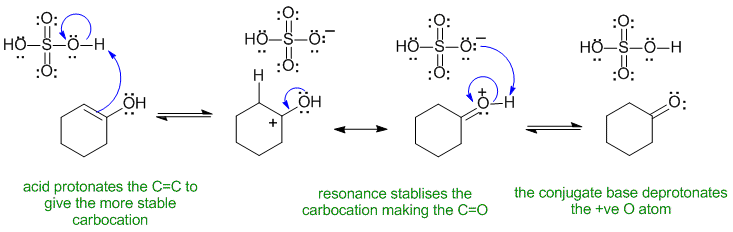
Common errors : drawing the reaction under basic conditions instead of acidic
B1
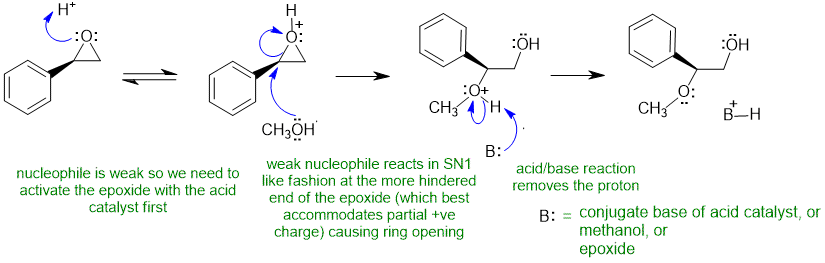
Common errors : drawing the reaction under basic conditions instead of acidic (i.e. using CH3O-), wrong regiochemistry, incorrect management of stereochemistry, using a base that was not present (read the question)
B2
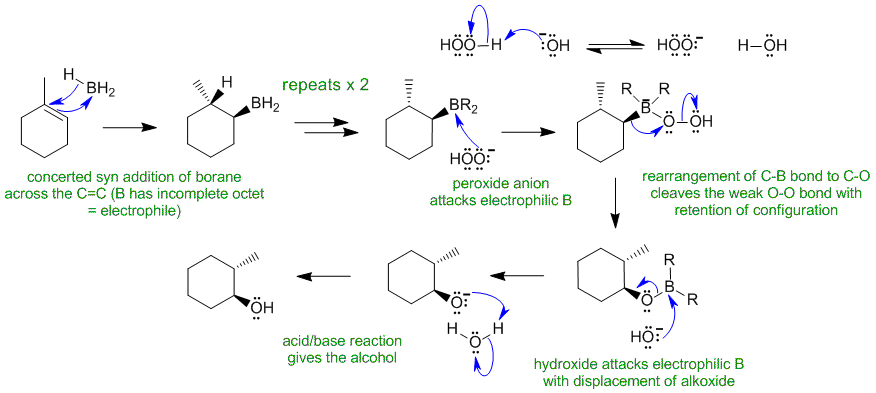
Common errors : not showing the stereochemistry, not showing a concerted addition (first step), showing B as having a lone pair, showing dissociation of the BH3 system. Note that the generally the easier concerted first step was done more poorly than the more complex oxidation reaction.