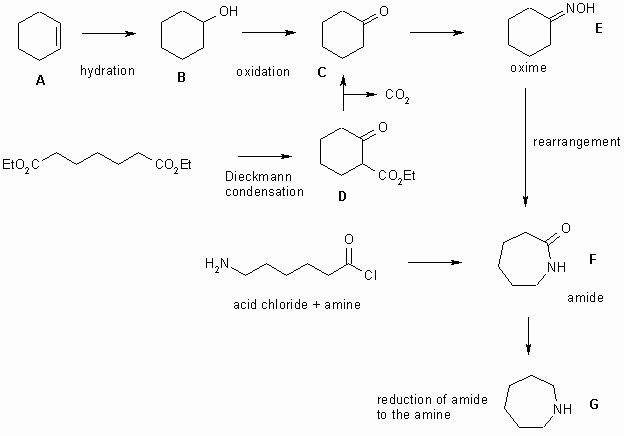
Work on the complete "flow chart" is in progress, in the mean time here are the answers....

The mechanism of E to F is actually an example of a Beckmann rearrangement (oxime to amide). Given that the structure of F should be obvious from the cyclisation of the 6-aminohexanoyl chloride, you should be able to suggest the curly arrows using the basic rules of mechanisms... hint, -OH group will get protonated to make a better LG....
A scheme will be added as soon as it's
available !