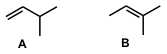
a. Elemental analysis : Since the % add up to 100% we know we have a hydrocarbon. The question tells us the mass spectrum showed the molecular ion at m/z =70, so the MW=70 g/mol. Hence one can calculate the molecular formula = C5H10
b. Index of hydrogen deficiencies = IHD = 1
c. A and B are acyclic (i.e no rings) so the IHD=1 implies we are looking at alkenes. The spectral data tells us that A has 5 H types and 4 C types and B has 4 H types and 5 C types (hint B has a C without any H attached). Note that in A the two H in the H2C=C are not equivalent as one is cis to the isopropyl group and the other is trans.
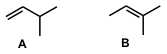
d.

e. We can determine which of A and B are more stable by comparing their heats of formation or combustion. Since the heat of formation of B is more exothermic than A, then B must be more stable than A.
Common errors:
General: Breaking valence rules (too many bonds, incorrect charges etc.). Incorrect structural representations such as when using line diagrams or being inconsistent when showing H atoms (i.e. showing some but not all)
a . Could not do elemental analysis calculations.
c. Did not know what acyclic meant and so drew cyclic structures.