| Chapter 1: Structure Determines Properties |
| Chapter 1: Structure Determines Properties |
Pushing Electrons and Curly Arrows
"Curly arrows" or "curved arrows" are how organic chemists communicate
and "think" about mechanisms.
The ability use curly arrows is probably the single most important skill or tool
for simplifying organic chemistry.
Understanding how to use curly arrows allows you to appreciate how organic chemistry works since the arrows show how reactions proceed and this helps remove the need
to memorise reactions.
Curly arrows show how the electrons and therefore how the bonds are reorganised. Reorganising bonds implies a reaction has taken place.
Curly arrows should "talk to you" ! Looking at a set of curly arrows literally tells you all the bonding changes, both breaking and forming that happen in a particular step of a reaction sequence.
| Study Tip: Many students struggle with organic chemistry because they never master curly arrows and so miss out on the important information they are trying to tell you. |
Steps to mastering curly arrows![]()
| |
|
|
| used to show the motion of pairs of electrons (more common) |
used to show the motion of single of electrons (i.e. radical reactions) |
We will focus on the more common
arrows ![]() here
:
here
:
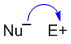 |
EXAMPLE
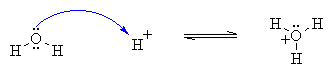 |
Arrow begins at a
lone pair on the O atom and goes to the H atom forming
a new O-H bond. |
 |
 |
 |
||
| |
|
Note: How do you know how much to include in a "step" ? The most common mistake students tend to make is that they merge several steps in to a single step.
This seemingly simple question is actually not easy to answer. The following factors should be considered:
Study Tip: REMEMBER Understanding the location of electrons and being able to draw the curly arrows that depict the mechanisms by which a reaction occurs is one of the most critical tools for learning organic chemistry since they allow you to appreciate what controls reactions, how reactions proceed and highlight the similarities between seemingly unrelated reactions. In a correctly drawn MECHANISM, curly arrows should be used to show ALL the BONDING changes that occur. |
 |
© Dr. Ian Hunt, Department of Chemistry, University of Calgary |  |