| Chapter 2 : Alkanes |
| Chapter 2 : Alkanes |
Bonding in H2
In Chapter 1 we introduced the idea that electrons have wave-like properties that led to the concept of electrons being in atomic orbitals (review ?) But we also need to consider what happens to the electrons and the associated atomic orbitals when a molecule forms.
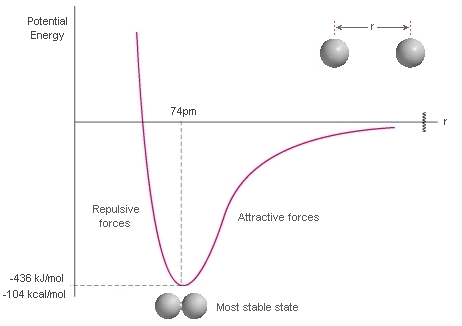 |
First, let's consider what happens to the energy
of two H atoms as they get closer to each other from infinite separation.
The attraction of the electrons to the nuclei of the other atom stabilises the system until they get so close that the nuclei start to repel each other. The most stable (lowest energy) state corresponds to the equilibrium H-H bond length 74pm. The energy difference between the most stable state and the energy at infinite separation (zero by definition) corresponds to the H-H bond strength. |
Now let's
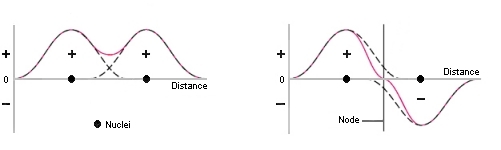
consider how the 2 electron
waves can interact with each other when the 2 atoms interact:
 |
|
| "in-phase" combination Note how the wave function is enhanced in the region between the two nuclei |
"out-of-phase" combination Note how the wave function is reduced in the region between the two nuclei |
In the in-phase combination there
is an increase in the electron
density in the internuclear region. This corresponds to the way
in
which we think of a covalent
bond : a sharing of a pair of electrons between two nuclei.
| © Dr. Ian Hunt, Department of Chemistry |