| Chapter 4: Alcohols and Alkyl Halides |
| Chapter 4: Alcohols and Alkyl Halides |
Reaction of Alcohols with Hydrogen Halides
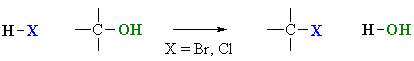
Summary
| SN1 MECHANISM
FOR REACTION OF ALCOHOLS WITH HBr |
|
| Step 1: Step 2: Step 3:
|
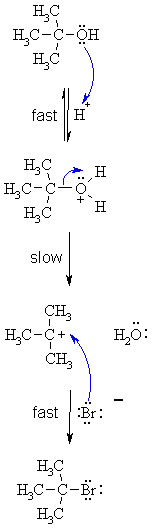 |
| © Dr. Ian Hunt, Department of Chemistry |