| Chapter 8: Nucleophilic Substitution |
| Chapter 8: Nucleophilic Substitution |
Nucleophilicity and basicity are very similar properties in that species that are nucleophiles are usually also bases (e.g. HO- , RO-).
This is not too surprising since in the LEWIS sense, they are functioning as LONE PAIR donors (i.e. both are LEWIS BASES), compare the two pairs of reactions mechanisms shown below to convince yourself.
However, it can avoid confusion by keeping these two types of reactivity separated because there are important differences....
1. Nucleophilicity:
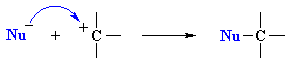
|
Both these reactions depict a nucleophile reacting with an electrophilic C atom |
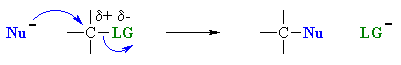 |
Kinetically controlled reactions of lone pair donor with
an electrophilic carbon (usually) atom resulting in the formation a new C-X
bond.
| |
Both these reactions depict a base reacting with an electrophilic H atom, a proton |
Equilibrium (thermodynamic) reaction of the lone pair donor with a proton, forming a new H-X bond
The following general reaction mechanisms show you why it
is important to appreciate the differences, since an anion that is reacting
as a nucleophile will result in a substitution, but if it reacts as a base,
then elimination will result, be it on a carbocation (SN1 vs E1)
or with the loss of a leaving group (SN2 vs E2).
| Substitutions | 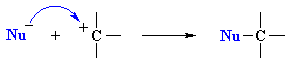 |
SN1 |
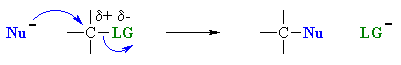 |
SN2 | |
| Eliminations | 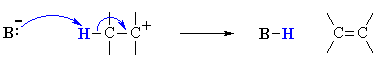 |
E1 |
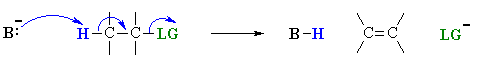 |
E2 |
| © Dr. Ian Hunt, Department of Chemistry |