| Chapter 15: Alcohols, Diols and Thiols |
| Chapter 15: Alcohols, Diols and Thiols |
Hydration of Alkenes
(review of Chapter 6)
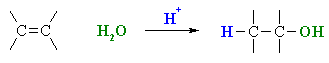
Summary
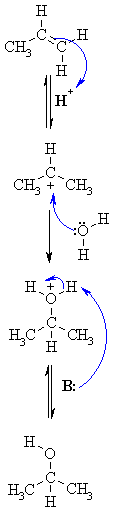
| MECHANISM
FOR REACTION OF ALKENES WITH H3O+ |
|
| Step 1:
An acid / base reaction. Protonation of the alkene to generate the more stable carbocation. The p electrons act pairs as a Lewis base. |
|
| Step 2:
Attack of the nucleophilic water molecule on the electrophilic carbocation creates an oxonium ion. |
|
| Step 3:
An acid / base reaction. Deprotonation by a base generates the alcohol and regenerates the acid catalyst. |
|
| © Dr. Ian Hunt, Department of Chemistry |