 |
Chapter 18: Enols
and Enolates
|
 |
Advanced Aldol Condensations
The more advanced topics are : Dehydration,
Mixed and Intramolecular Aldol reactions.
Dehydration
of Aldol products
The dehydration of the initial Aldol
products can occur, it is often favoured by the following factors:
- The Aldol product must
still have an α-hydrogen between the carbonyl and
the -OH group.
- Look for extended
conjugated in the product (i.e. increased stability).
- Heating the reaction often favours
elimination of water (dehydration).
- Non-aqueous reaction conditions
favour the removal of water (equilbrium shifts to dehydration products).
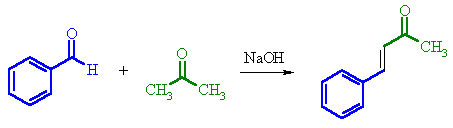
Mixed
Aldols
A mixed Aldol reaction means
that the enolate and the carbonyl are not from the same aldehyde or ketone.
The most useful mixed Aldols are
those where:
- Only one reactent can form an
enolate.
- This means the other reactent
lacks α-hydrogens (for examples, see below) and
therefore must be the electrophilic carbonyl component.
- One
reactent is more electrophilic than the other and functions as the electrophilic
carbonyl component.
- Recall
that in general, aldehydes are more reactive (electrophilic) than ketones
and will usually be the electrophile.
Otherwise there are selectivity problems
that give rise to complex mixtures of products.
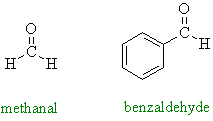 |
|
Neither of these
aldehydes has α-hydrogens and therefore they
cannot form an enolate. |
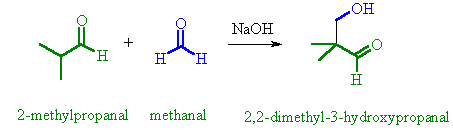 |
|
Only one of this
pair of aldehydes has an α-hydrogen |
QUESTIONS
| |
In the above example, which aldehyde forms the enolate ?
|
ANSWER |
| |
Why can't the aldol product dehydrate ?
|
ANSWER |
Intramolecular
Aldols
- Dicarbonyl compounds can be used
to give intramolecular Aldol reactions.
- This means the enolate component
and the carbonyl component are parts of a single larger molecule.
- The selectivity is typically
controlled by the stability of the cyclic Aldol product.
- This favours the formation of
the more stable 5- or 6-membered rings (review)
- To work out the best product,
check the permutations that are possible and look for the favourable ring
sizes.
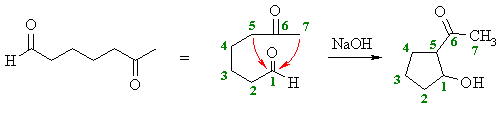 How can you deduce this product ?
How can you deduce this product ?
- Since aldehydes are more electrophilic than ketones, the aldehyde will be the electrophile and the ketone the nucleohile (i.e. the enolate) (see above)
- There two possible enolates (at
C5 or C7)
- The more favourable ring size
will the 5-membered ring.
Study Tip:
When doing intramolecular reactions, it is a good idea to count carbons
to avoid silly errors.
If you start numbering at the electrophilic C=O C atom, then it will "show you" the rings sizes... |



