| Chapter 22: Amines |
| Chapter 22: Amines |
Reduction of Amides
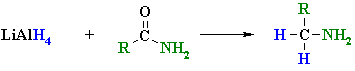
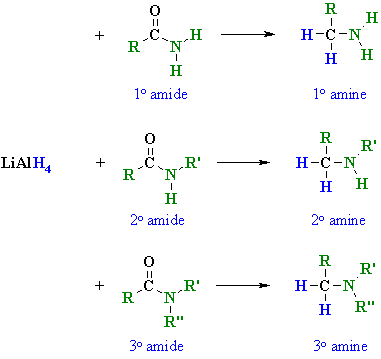
Summary
|
|
|
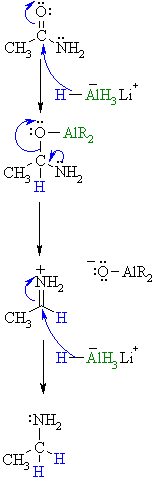
| Step 1: The nucleophilic H from the hydride reagent adds to the electrophilic C in the polar carbonyl group of the ester. Electrons from the C=O move to the electronegative O creating an intermediate metal alkoxide complex. |
|
| Step 2: The tetrahedral intermediate collapses and displaces the O as part of a metal alkoxide leaving group, this produces a highly reactive iminium ion an intermediate. |
|
| Step 3: Rapid reduction by the nucleophilic H from the hydride reagent as it adds to the electrophilic C in the iminium system. π electrons from the C=N move to the cationic N neutralising the charge creating the amine product. |
|
| © Dr. Ian Hunt, Department of Chemistry |