|
|
|
|
Reduction of Esters
(review of Chapter 15)
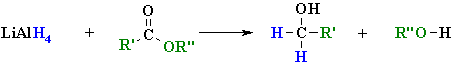
Summary
|
|
|
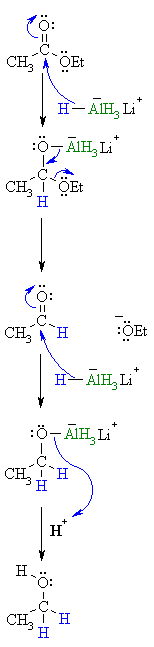
| Step 1: The nucleophilic H from the hydride reagent adds to the electrophilic C in the polar carbonyl group of the ester. Electrons from the C=O move to the electronegative O creating the tetrahedral intermediate a metal alkoxide complex. |
|
| Step 2: The tetrahedral intermediate collapses and displaces the alcohol portion of the ester as a leaving group, in the form of the alkoxide, RO-. This produces an aldehyde as an intermediate. |
|
| Step 3: Now we are reducing an aldehyde (which we have already seen) The nucleophilic H from the hydride reagent adds to the electrophilic C in the polar carbonyl group of the aldehyde. Electrons from the C=O move to the electronegative O creating an intermediate metal alkoxide complex. |
|
| Step 4: This is the work-up step, a simple acid/base reaction. Protonation of the alkoxide oxygen creates the primary alcohol product from the intermediate complex.
|
|
| © Dr. Ian Hunt, Department of Chemistry |