| Chapter 1: Structure Determines Properties |
| Chapter 1: Structure Determines Properties |
| All matter is
composed of atoms. Atoms are an ordered
collection of various subatomic particles. The central nucleus
contains the protons and neutrons, around
this are the all
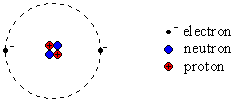
important electrons. The simple schematic diagram to the left shows this simple model of a helium atom. The nucleus contains two protons and two neutrons, and then there are two electrons. You should be familiar with
the subatomic particles and how to
determine how many of each a specific atom has. |
 |
In introductory organic chemistry courses, the majority of the compounds you will encounter are based on a limited selection of atoms : the atoms of greatest interest are H, C, N, O, and the halogens F, Cl, Br, I plus a few others such as Li, Na, Mg, Al, B, P, S. Later, when one probes deeper into more advanced synthetic methods, one needs to be familiar with many more elements.
Try the questions about atoms. If you are not comfortable with these you should look at a bit more detail.
| © Dr. Ian Hunt, Department of Chemistry |