| Chapter 6: Reactions of Alkenes: Addition Reactions |
| Chapter 6: Reactions of Alkenes: Addition Reactions |
Halogenation of Alkenes
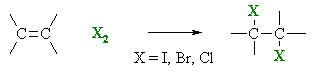
Summary
| These 3D JSMOL images show the structure of
the bromonium ion formed during the reaction of ethene with Br2. Note how the Br is attached to both the C atoms.
QUESTION : What is the formal charge on the bromine atom ? ANSWER When the Br- nucleophile attacks, it order to reach the C atoms, it will
attack from the least hindered face, the side opposite to the large Br in
the intermediate. |
Related reactions
| MECHANISM FOR REACTION OF ALKENES WITH HALOGENS | |
| Step 1: The π electrons act as a nucleophile, attacking the bromine, displacing a bromide ion but forming a cationic cyclic bromonium ion as an intermediate. |
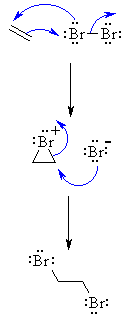 |
| Step 2: Attack of the nucleophilic bromide from the side away from the bromonium center opens the cyclic bromonium ion to give overall anti addition. |
|
| © Dr. Ian Hunt, Department of Chemistry |