| Chapter 16: Ethers, Epoxides and Sulfides |
| Chapter 16: Ethers, Epoxides and Sulfides |
Cleavage of Ethers by HI or HBr
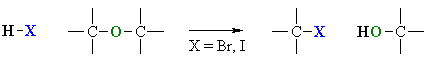
Reaction type: Nucleophilic Substitution
Summary
QUESTIONS
| ANSWER | ||
| ANSWER |
Related reactions
|
|
|
| Step 1: An acid/base reaction. Protonation of the alcoholic oxygen to make a better leaving group. This step is very fast and reversible. The lone pairs on the oxygen make it a Lewis base. |
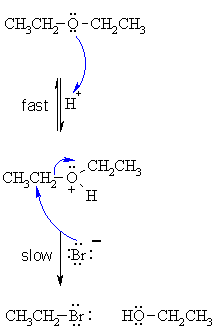 |
| Step 2: The bromide ion functions as the nucleophile and attacks to displace the good leaving group, neutral alcohol molecule, by cleaving the C-O bond. This results in the formation of an alkyl bromide and an alcohol. |
|
| © Dr. Ian Hunt, Department of Chemistry |