| Chapter 20: Carboxylic Acid Derivatives. Nucleophilic Acyl Substitution |
| Chapter 20: Carboxylic Acid Derivatives. Nucleophilic Acyl Substitution |
Reactions of Nitriles
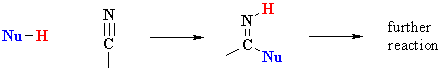
Overview
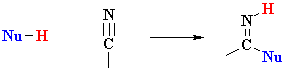 versus
versus 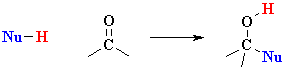
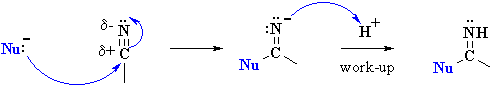
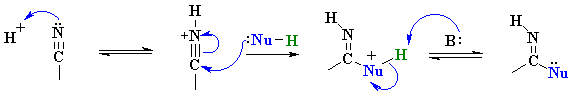
Examples of such nucleophilic systems are : H2O, ROH
| The protonation of a nitrile gives a structure that can be redrawn in another resonance form that reveals the electrophilic character of the C since it is a carbocation. | 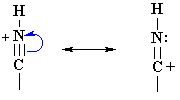 |
| © Dr. Ian Hunt, Department of Chemistry |