| Chapter 21: Ester Enolates |
| Chapter 21: Ester Enolates |
In this chapter we look at the chemistry
of enolates of esters and active methylene systems.
Recall that we have previously looked at the enolates
of aldehydes and ketones.
What we will see here is analogous to that material and can be treated in a
very similar fashion.
Before continuing, you should make sure you are familiar with the reactions where the esters are attacked by nucleophiles due to the electrophilic character of the carbonyl C and undergo nucleophilic substitution (Chapter 20)
All that is going to change in this chapter is the nature of the attacking nucleophile...
| Treatment of esters with a
suitable base can lead to the formation of a nucleophilic
species called an enolate that will react with an electrophile.
These C nucleophiles are useful for making new carbon-carbon bonds. |
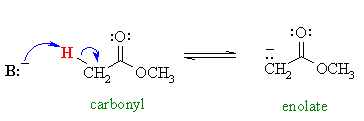 |
Enolates
| © Dr. Ian Hunt, Department of Chemistry |