Here is an post-mortem analysis / "how to" for the MT. The questions are split by the sections. At the start of each section are a few suggestions of what to look for or how to tackle the question type.
RELATIVE PROPERTIES:
Identify the controlling feature, which is not
always as obvious as it may appear. Look for two pairs of similar systems to compare
that have minimal differences in structure. If a compound is named, draw it out.
If a reaction is involved, identify the type of reaction and then what the controlling
factors are.
Qu1: D
Reaction is the hydrogenation of either alkene or alkynes or aromatics. The reactivity towards reduction is determined by the strength of the pi bonds: the weaker the pi bond, the faster the reaction. So alkynes ii reduce more rapidly than alkenes iii. Aromatic C=C i reduce more slowly than alkene C=C due to the aromatic stabilisation. So ii > iii > i.
Qu2: D
Benzylic hydrogens are found on the C atoms attached to benzene rings. So i has 0, ii has 4 x 3 = 12 and iii has 2. Hence ii > iii > i.
Qu3: C
Experimental yields.... First you need to balance the reaction equation and then work out the moles of each reagent used to determine the limiting reagent:
3 H2C=CH2 + BH3--[work up] --> 3 CH3CH2OH |
|||
ethene |
borane |
ethanol |
|
| MW (g/mol) | 28 |
15 |
46 |
ii 2.8 g ethene = 0.1 mol, due to the stoichiometry borane is the limiting reagent (0.02 mol) and we can get 3 x 0.02 mol of alcohol. We get 2.3g of alcohol = 0.05 mol, so yield = 0.05 / 3 x 0.02 = 83%
iii ethene = 0.3 mol, due to the stoichiometry there is excess borane so the alkene is the limiting reagent and we can get 0.3 mol of alcohol. We get 0.1 mol of alcohol so yield = 0.1 / 0.3 = 33%
So ii > i > iii.
Qu4: C
Reaction is the Diels-Alder reaction and we are looking at the dienes reacting with the dienophile but-3-en-2-one (drawn in the question). The reactivity increases in the Diels-Alder reaction with electron donating groups on the diene and the diene needs to be able to adopt an s-cis conformation. i is the simplest conjugated diene 1,3-butadiene, it has conformational flexibility ii has an alkyl group substitution and is locked s-cis, and iii can not achieve the reactive s-cis conformation because the ring locks it in the unreactive s-trans conformation. Therefore the reactivity is ii > i > iii.
Qu5: AB
The reaction is the hydroboration / oxidation of alkenes. First draw out the starting material, 1-methylcyclopentene. ii and iii have correctly added the HO and H across the C=C therefore i has the lowest yield. ii is the Markovnikov product, while iii is the anti-Markovnikov product. Since hydroboration / oxidation gives the anti-Markovnikov product as the major product then we get iii > ii > i.
Qu6: C
All about enantiomeric excess and optical rotation. For i based on the masses of the two enantiomers drawn, the e.e. is 33% (use (1.0 - 0.5)/(1.0 + 0.5)). For ii a little more complex, but work out the e.e. via the rotation... [a]D (sample) = a / cl = +1.27 / (1.0/10) = 12.7. Therefore e.e. = 100%. And for iii a racemic mixture has an e.e. of 0 (by definition). So ii > i > iii.
Qu7: E
The reactions of alkenes and alkynes with aq. acid are controlled by carbocation stability and the number of alkyl groups on the pi unit (more alkyl groups makes them more electron rich and hence more nucleophilic). Alkynes are less reactive since they would require the much less favourable termolecular pathway to avoid the formation of the very unfavourable vinyl carbocations. Therefore iii > i > ii.
Qu8: C
Carbocation
stability.... due to (a) alkyl groups, which are weak electrons donors, and (b) resonance with Br lone pairs. These effects add stability due to charge delocalisation. i is secondary with an Br attached that can stabilise the charge via resonance using the Br lone pair, ii is a secondary allylic carbocation and iii is an unfavourable vinyl carbocation. Hence ii > i > iii.
Qu9: A
The relative stability of systems.... in this case we are looking at polyenes and the degree of alkene substitution. i and ii are both conjugated dienes whereas iii is an isolated diene. Conjugated dienes are more stable than isolated dienes. The difference between i and ii is that i has a tetrasubstituted C=C while ii only has a disubstituted so i is more stable than ii. Therefore we get i > ii > iii
Qu10: E
All the bonds are CH bonds. The factors involved are (1) the hybridisation of the C atom and (2) allylic systems. In terms of hybridisation, sp3 C-H bonds are weaker then sp2 C-H due to the increased s character in the sp2 situation creating a shorter and stronger bond. Allylic C-H bonds are weaker due to the proximity of the electron rich pi bond. In i we have a secondary sp3 system. In ii is secondary sp3 system that is allylic and in iii a vinyl C-H i.e. sp2C.. So in terms of C-H bond strengths: iii > i > ii.
STARTING MATERIALS AND PRODUCTS:
If you are trying to find the product, then
you should probably just work forwards through the sequence of reactions.
If you are looking for the starting material,
then working backwards is probably the best way to go....
Basically depends on the need to know and identify
the reactions, this is often triggered by looking at the functional groups in
the molecules.
Qu11: B
Working forwards... The alkyne is converted to the nucleophilic terminal acetylide by using the NaNH2 then reacted in an SN2 fashion with the epoxide to make but-3-yn-1-ol, HCCCH2CH2OH. This is then followed by hydration of the alkyne to give a methyl ketone alcohol, B.
Qu12: A
Working backwards.....reactions look like alkene ozonolysis with a reductive work-up and therefore reconnect the carbonyls to reveal ethylcyclopentene where the alkene has been formed by an E2 elimination of an alkyl halide (the base/heat suggests this) where the alkene includes the C where the alkyl chain attaches to the ring = A
Qu13: D
Working forwards... the catalytic hydrogenation (a reduction) of an alkyne to an alkene using Lindlar's catalyst gives propene that then undergoes radical addition of HBr to the alkene giving the anti-Markovnikov product, 1-bromopropane, CH3CH2CH2Br.
Qu14: E
Working forwards.....addition of HBr to the alkene giving the Markovnikov product, 2-bromo-2-methylbutane, (CH3)2CBrCH2CH3 followed by an E2 elimination of an alkyl halide (the base/heat suggests this) according to Zaitsev's rule (gives the more highly substituted, more stable alkene) E.
Qu15: A
Need to eliminate the alcohol to make an alkene and then do an allylic radical bromination. Since none to the reaction conditions have acid to eliminate the alcohol, first need to convert the -OH to a halide, then use smaller base to do an E2 elimination of an alkyl halide in accord with Zaitsev's rule. B step 3 would add HBr to the alkene not an allylic radical substitution. C would not have an alkene as the product. D would not form an alkene because alcohols are eliminated with acid. E would form the anti-Zaitsev alkene and then add HBr across that alkene to give 2-bromobutane.
Qu16: D
Working forwards.... looking at the alkyne starting material, we need to add 2 C atoms via an alkylation and reduce the alkyne to a cis-alkene. The alkyne is first converted to the nucleophilic terminal acetylide by using the NaNH2 then reacted in an SN2 fashion with ethyl halide (CH3CH2X) to give 2-pentyne. Catalytic hydrogenation (a reduction) of an alkyne to an alkene using Lindlar's catalyst gives cis-pent-2-ene.
Qu17: B
Working forwards... bromination of the alkene gives the 1,2-dibromide followed by double elimination (- 2 HBr) with the strong base will give the alkyne (note the position of H available for elimination dictates this outcome). The original location of the C=C dictates the structure.
Qu18: E
Working forwards.. the alkene and the alcohol in the starting materials mean that the reactions look like intramolecular alkoxymercuration / demercuration of an alkene (note that no water nor a new alcohol is being added) - this would result in a cyclic ether as the product. Counting / numbering C atoms will reveal the exact structure. B would have been the product if water had been present (Markovnikov product).
REGIO- and STEREOCHEMISTRY:
How well do you know your reagents ? Look
at what has actually happened in terms of the reaction functional group transformation
and then first look for any regiochemical issues then finally the stereochemistry
last (it's the hardest to sort out). In cases where more than one product
is formed in equal amounts (e.g. the enantiomers), then both must be selected
for full marks, part marks are given when only one of the pair is selected.
Advice : in each case draw the starting material in the
conformation in which is reacts or the product in the conformation in which
it is initially formed using wedge-hash diagrams. It is a good idea to draw
the materials in such a way that the new bonds are in the plane of the page.
Once you have drawn the materials it may also be good for you to use model kits
for these questions too. Once you have drawn the materials in this way,
you may need to consider rotations around sigma bonds to make your answer match
the options. An alternative approach could be to assign configurations
to your drawn answer to compare them with the options - this can be slow and
prone to error.
Qu19: B
Working forwards...the starting material is an alkyne, the first set of reagents indicate
the dissolving metal reduction to give the trans-alkene. The alkene is then reacted with cold alkaline potassium permanganate to give a 1,2-diols via a syn addition - hence not C, D or E (wrong functional groups). The syn addition of the diols to the trans-alkene will give B rather than the meso compound (A). Darn those Fischer projections!
Qu20: E
Working forwards...the starting material is an alkene, which reacts with the aqueous chlorine to give the 1,2-chlorohydrin via an anti addition, where the -OH ends up at the more substituted (tertiary) position due to the +ve character of the intermediate halonium ion. A has the functional groups. B has the wrong stereochemistry. C and D have the wrong regiochemistry.
Qu21: C
Working forwards, it looks like we have done an anti-Markovnikov (Br at the less substituted end of the alkene) radical addition of HBr to the alkene. The required alkene was formed by the carbocation rearrangement of due to protonation of an alkene (follows Zaitsev's rule). A doesn't have enough C atoms, B lacks any functional group to react with either of the sets of reagents and neither D nor E would react with acid to eliminate to give an alkene (alkyl chlorides are eliminated using base / heat).
Qu22: C
Working forwards, treatment of a 1,2-halohydrin with sodium hydroxide as a base forms the epoxide via an SN2 reaction. This means that we need to set up the starting material in the conformation in which is reacts where the HO and the Br are anti to each other so that the Nu can attack the backside (at 180 degrees to the LG) and cause the normal SN2 inversion. A, D and E all have the wrong functional groups. B has the wrong stereochemistry because you've ignored the SN2 requirements.
Qu23: B
Working backwards... the reaction is the hydroboration-oxidation of an alkene to give 3-methylpentan-2-ol. The reaction gives the anti-Markovnikov alcohol via a syn addition due to the concerted addition of the B and H across the C=C. Note that the -OH is formed with retention of stereochemistry when the B atom is replaced. Redraw one of the enantiomers so the -OH and the H that in the conformation in which they are added (i.e. syn) then "remove" them to reveal the C=C unit. A gives 3-methylpentan-1-ol, C and E would give 2-ethylbutan-1-ol. D has the wrong stereochemistry.
Qu24: D
This is a Diels-Alder reaction.... hints are the dienophile (qu25) and dienes (qu24). The challenge here is to realise that the C=C has been removed by the catalytic hydrogenation... the only way to recognise where it was it to recall that it is opposite in the cyclohexane unit to the location of the dienophile which is easily recognised by looking at the ester groups. With the C=C back in place, then you can push the curly arrows to "reverse" the Diels-Alder and reveal the diene and the dienophile.
In qu 24, the process described above reveals that we need the simplest diene, 1,3-butadiene.
Qu25: C
As per qu 24, the process reveals that we need the trans-diester, C. D has the wrong stereochemistry and E would have given cis ester groups in the product after the catalytic hydrogenation
PI SYSTEMS:
All about knowing properties of pi systems such as resonance, structure, bonding, shapes, and terminologies etc.
Qu26: AB
Conjugation requires an extended pi system (at least 3 atoms) where the pi systems can interact
(parallel, not perpendicular). This is not restricted to all carbon systems.
Make sure you draw out all the bonds to be sure. A has conjugated C=C and C=O an isolated. B is a conjugated diene. C and D are isolated dienes (sp3 C between the 2 C=C units). E is also an isolated type of system.
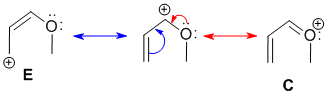
Qu27: CE
Resonance contributors are derived via the delocalisation of the pi electrons with the pi system and can be derived by pushing curly arrows. Use the rules for recognising resonance structures. A, B and E can be ruled out because the +ve charge can't be located on a C that was sp2 in the original structure.

Qu28: D
The key facts here are (1) conjugated dienes are more stable than isolated dienes (due to conjugation stabilisation), (2) in acyclic systems, trans alkenes are more stable than cis (sterics) and (3) in conjugated dienes the s-trans is more stable than the s-cis (sterics). Therefore the most stable system is D where we have an conjugated trans alkenes with a s-trans conformation. A and C are conjugated cis alkenes. B and E are isolated dienes.
Qu29: B
The most exothermic heat of hydrogenation means we are looking for the least stable isomer. The key facts here are (1) alkyl groups stabilise alkenes or alkynes so more substituted systems are the more stable, (2) alkynes are less stable than conjugated or isolated dienes. (3) branched alkyl groups are more stable than straight chains. Therefore the least stable system is B the terminal alkyne with the unbranched alkyl substituent. A and E are dienes. C has a branched alkyl substituent and D is an internal (disubstituted alkyne).
Qu30: B
Resonance energy is the amount of stabilisation caused by resonance. In these polyene examples, it will be the one with the most conjugation. B is a conjugated triene.
Qu31: AC
Tautomers (remember alkyne hydration ?) of ketones are the enols. B is a resonance contributor. D not even an isomer (it's a reduction product). E is a conjugate base of the enol.
Qu32: E
Reaction is the hydrogenation of either alkene or alkynes or aromatics. The reactivity towards reduction is determined by the strength of the pi bonds: the weaker the pi bond, the faster the reaction. So alkynes reduce more rapidly than alkenes and aromatic C=C reduce more slowly than alkene C=C due to the aromatic stabilisation.
Qu33: C
The E isomer requires that the higher priority groups at each end of the C=C (based on the Cahn-Ingold-Prelog rules: Cl > CH3 and Et > H) are on the opposite sides = C. A is Z, B and D are 4-chloropent-2-ene. E is (Z)-3-chloropent-2-ene.
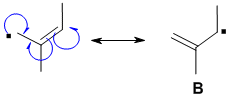
Qu34: B
Resonance contributors are derived via the delocalisation of the pi electrons with the pi system and can be derived by pushing curly arrows. Use the rules for recognising resonance structures. A, C and D can be ruled out because radical center has moved to C that were sp3 to begin with so an H would have had to move. E is wrong because the methyl group has become =CH2.